Designing batteries for space exploration
The challenge of designing batteries for the Europa Lander mission
Welcome to Lithium Horizons! This newsletter explores the latest developments, companies, and ideas at the frontiers of energy materials. Subscribe below to get the next article delivered straight to your inbox.
“Space, the final frontier… to boldly go where no man has gone before.”
— Captain James T. Kirk, Star Trek
Just as Captain Kirk dreamed of exploring the unknown, engineers dream of powering spacecraft for unknown environments. Space exploration often involves missions to some of the universe’s most inhospitable regions, making robotic spacecraft essential. These spacecraft require reliable power sources, typically provided by energy generators such as photovoltaic solar arrays or radioisotope thermoelectric generators. However, energy generation alone is not always sufficient, particularly during peak power demands. For this reason, energy generators are usually paired with energy storage systems, such as batteries.
Batteries can be categorized as either primary or secondary. Primary batteries are designed for a single use, providing power without recharging, which makes them ideal for space missions where no additional energy generation or storage is available. Examples of planetary probes that used lithium–sulfur dioxide (Li–SO₂) primary batteries include the Galileo and Cassini spacecraft.
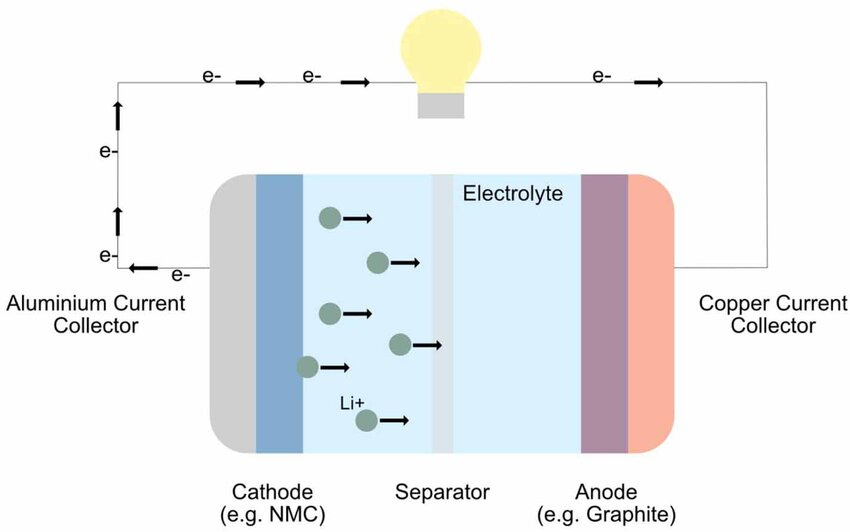
Conversely, secondary batteries are rechargeable (e.g., lithium-ion; Figure 1), allowing them to undergo multiple charge–discharge cycles. This capability makes them suitable for extended space missions that require long-term, sustainable energy storage. The European Space Agency's experimental Proba-1 Earth-observation mission in 2001 was the first to use rechargeable lithium-ion batteries in space.
What constraints do batteries face in extreme environments? How do they differ from those used in everyday applications such as smartphones or electric vehicles? How are they designed, and who manufactures them? These are some of the key questions addressed in this article.
We begin by examining the harsh conditions batteries must endure in space, followed by a brief history of batteries used in space missions, along with their performance requirements and design considerations. We conclude with a discussion of spacecraft designed for Europa missions and an overview of companies that manufacture batteries specifically for space applications.
Let's dive in! 🔋
NOTE: If you are reading this in your email client, the text may be clipped due to its length. Click on "View entire message" at the bottom of your email to read the whole article, or open it in your browser or Substack app.
Extreme conditions
Energy storage requirements for space exploration vary depending on the destination and the nature of the mission. Nevertheless, these systems must consistently withstand a range of severe environmental conditions such as extreme temperatures, radiation, microgravity, and vibration, all while providing sufficient power and energy output.
To appreciate the severity of these conditions, consider the planet Venus. Landing a rover on its surface would require an energy storage system capable of functioning at approximately 500 °C under about 90 atm of pressure. These conditions are roughly 20 times higher in temperature and 90 times greater in pressure than the standard operating limits of commercial lithium-ion battery systems.
Extreme pressures pose significant challenges for the structural design of battery packs. While moderate pressure can improve interfacial contact between the layers of a battery cell, excessive pressure can hinder ion transport, ultimately reducing battery cycle life.
Standard lithium-ion batteries operate optimally between 10 °C and 40 °C. Temperatures outside this range result in degraded performance. At low temperatures, increased electrolyte viscosity and slowed reaction kinetics raise internal resistance and elevate the risk of electrical shorts. At high temperatures, electrolyte degradation, separator melting, and eventual cathode decomposition can occur. The release of oxygen during cathode breakdown can initiate thermal runaway, leading to catastrophic battery failure.1
Beyond temperature and pressure, radiation is another critical factor. Earth's magnetosphere protects the planet from much of this harmful radiation, but once outside it, spacecraft are exposed to intense irradiation without adequate protection. Radiation exposure can alter material properties, leading to unpredictable and often degraded behavior (Figure 2).
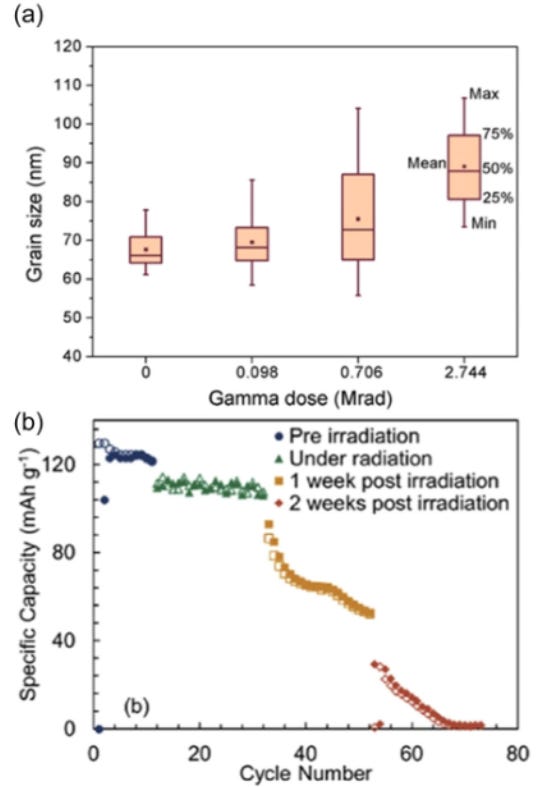
Ionizing radiation, such as gamma rays, can damage liquid organic electrolytes by generating free radicals that react with other components within the battery cell. Radiation can also induce cross-linking in polymer binders, compromising the structural integrity of electrodes. Additionally, it may alter the pore structure of polymer separators, affecting their wetting behavior. Neutron or ion irradiation induces defects in the crystal structure of materials. Collectively, these effects can reduce battery capacity, shorten cycle life, modify the solid-electrolyte interface, and increase internal cell resistance.
Mechanical stresses such as vibration, acceleration, and impacts are additional considerations, particularly during liftoff or landing. Vibration can compromise interconnections, including electrode-to-tab welds or terminal-to-busbar connections within a battery module or pack.
Understanding these environmental challenges helps explain why space battery development has always involved trade-offs between energy density, cycle life, and reliability.